Are you wondering what to do with leftover candy? You're in for a treat!
This experiment is sugar, and spice, and everything science! Join us as we dive into some chemistry (and our candy stash) and we'll give you a fun trick for what to do with your leftover treats.
We'll use a little bit of candy to make pictures that swirl like magic, to explore chemistry, and to practice making predictions and observations.
Materials you will need:
- Candy with a hard shell, like Skittles or M&M’s
- Warm water
- Shallow dish or plate that can hold liquid

Directions:
Step 1:
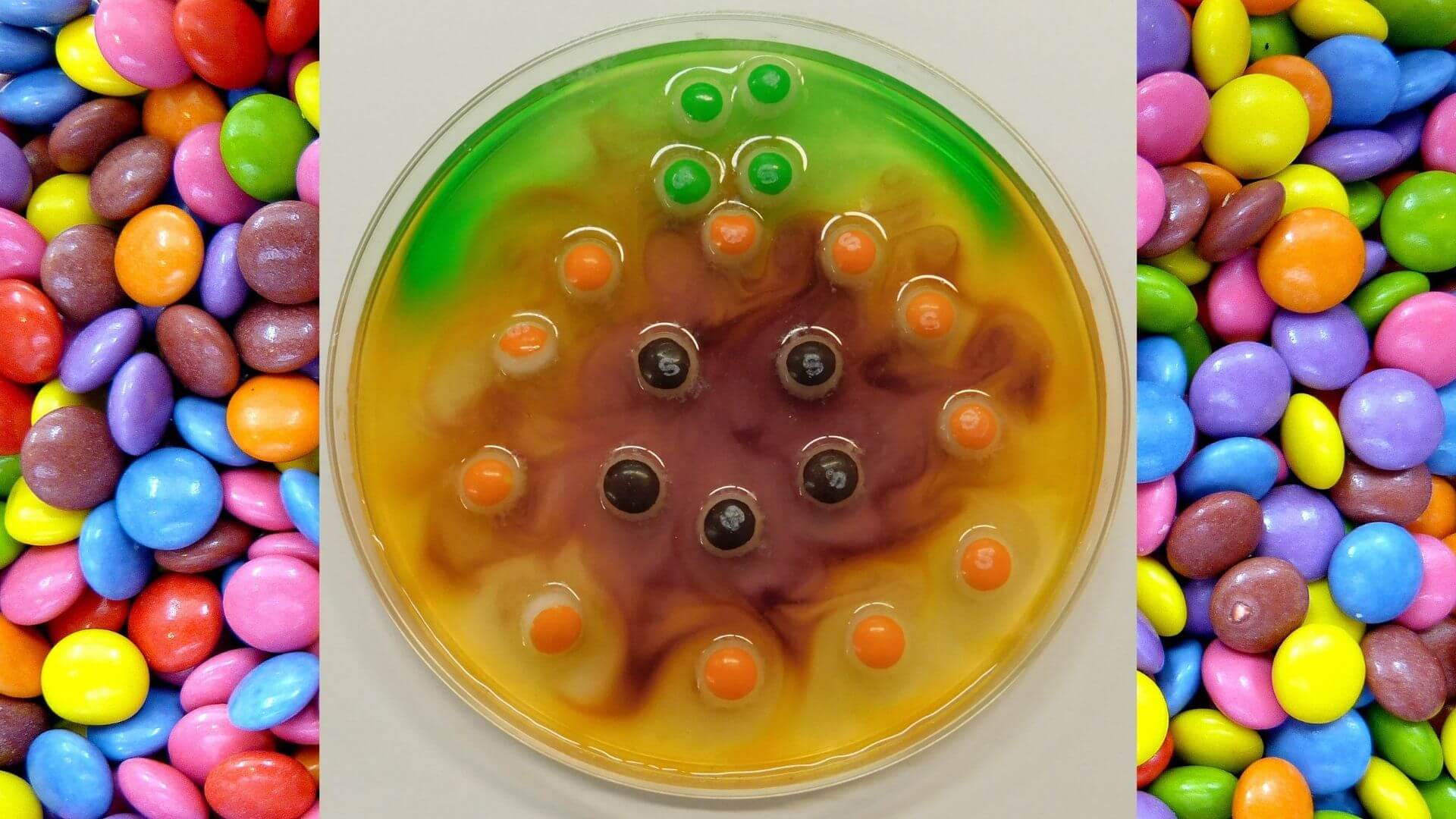
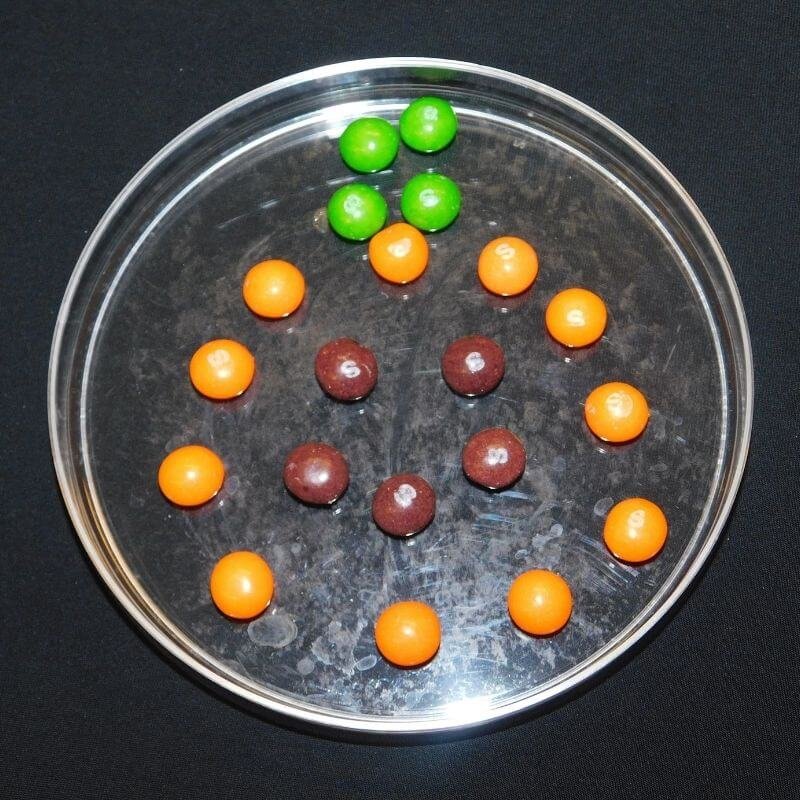
Arrange your candy in a design on your dish.
- You can try arranging them in a circle around the edge of the dish, or making pictures with them. Since we're using Halloween candy, we made a pumpkin.
Step 2:
Slowly pour your warm water over the candy.
Encourage your scientist to answer these questions:
- Before you add water, ask your scientist what they think will happen and why. This is called a hypothesis.
- What happens to the letter on the candy?
- Why do think the colors are moving?
- Why do you think the colors aren’t mixing?
- How do you think you could speed up the reaction

Step 3:
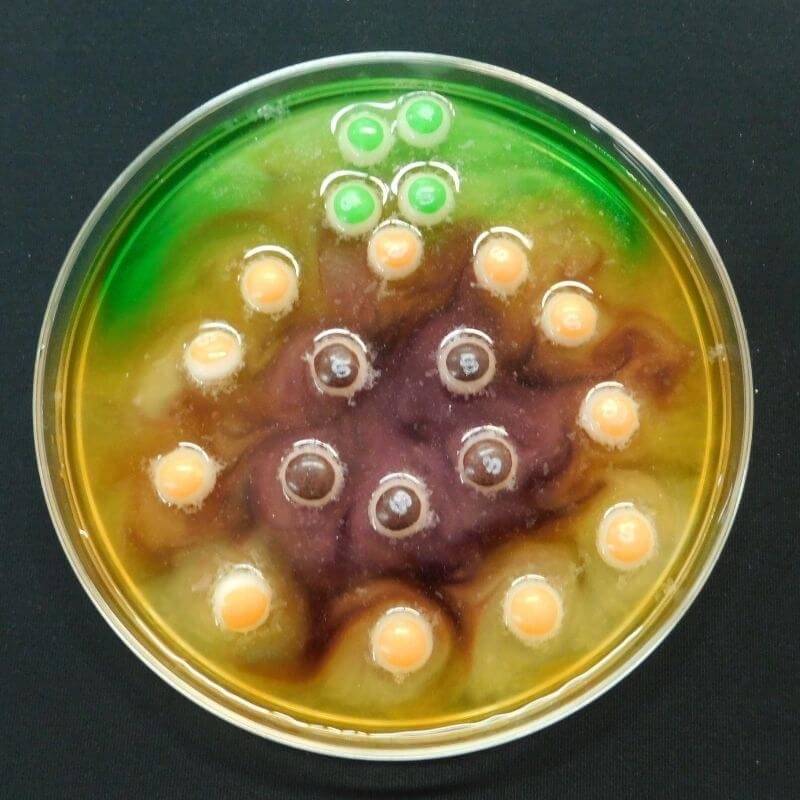
Watch what happens! What do you observe?
Make it sweeter!
- Make different designs. How are the color patterns different based on the design you make?
- Add another piece of candy after you’ve added water and the colors have started to spread out. What happens?
- Add a sugar cube to the candy after you’ve added the water and the colors have started to spread out. What happens?
- Experiment with different water temperatures. What temperature works best?
- Try using different candies. Which ones do you think will cause colors to spread out across the water
Expand on the Activity!
The Science
The colored shells on Skittles and M&M’s are made out of sugar and food coloring. As the sugar and food coloring dissolve in water, they diffuse (or spread out) across it. This changes the clear water to the colors of the candy.
The colors move from the area with the highest concentration of color (the candy and the area right next to it) to the area with lowest concentration (the area farthest away from the candy). Watch how the color moves away from the candies. Molecules moving from an area of higher concentration to an area of lower concentration is called a concentration gradient.
The colors don’t mix because of something called water stratification. Each color of food coloring has a slightly different chemical make-up. Because of this, they have slightly different densities. This keeps the colors from mixing as they spread out.
Try some more kitchen chemistry!
Tie-Dye Milk Experiment: Learn Chemistry in Your Kitchen
OSC At Home Emails
Get a round up of our latest activities and ideas delivered straight to your inbox so you don't miss a thing!
Support OSC At Home
In these ever-changing times, it is our pleasure to adapt quality Orlando Science Center experiences to engage with everyone while they are safe at home. Please consider supporting our operating fund to ensure we can continue developing resources today and well into the future. Thank you for your generosity and support!