Learning has never been sweeter with this kitchen chemistry for kids of all ages!
Everything we interact within our day-to-day lives is made out of molecules. There are countless different kinds of molecules, each made out of atoms of different elements.
This kitchen chemistry for kids will help build an understanding of atoms and molecules as we create our own atomic marshmallow models!
Materials you will need:
- Colored marshmallows
*If you don’t have marshmallows, you can use clay, playdough, etc... - Toothpicks

Molecules:
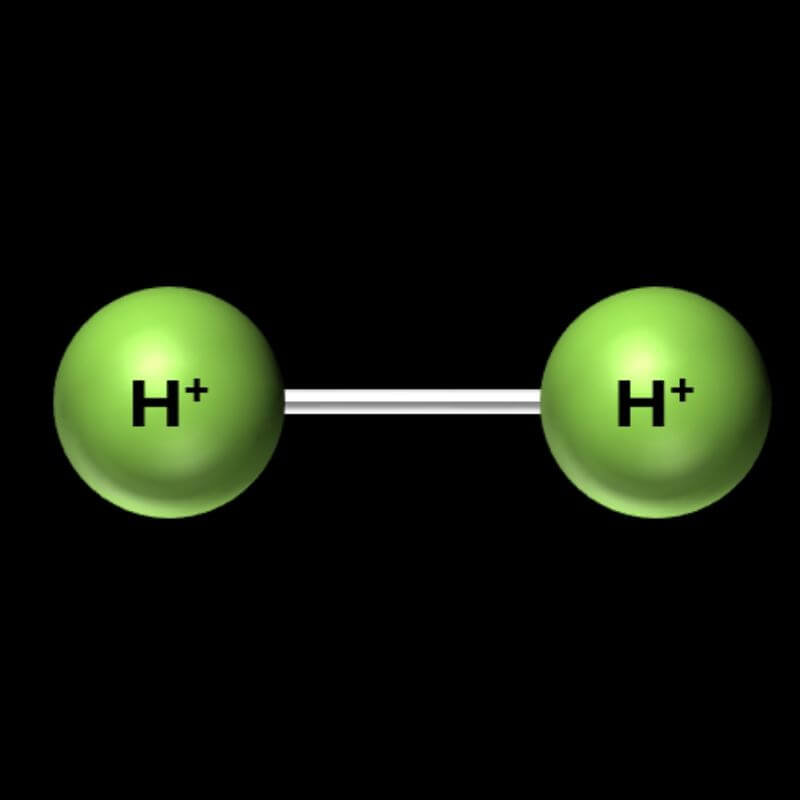
Hydrogen (H2):
- Some molecules are homonuclear, which means they are made up of just two atoms of the same element. Let’s make a homonuclear hydrogen molecule.
- To make a hydrogen molecule, grab 2 marshmallows of the same color. Then connect them with toothpicks, as shown in the picture.
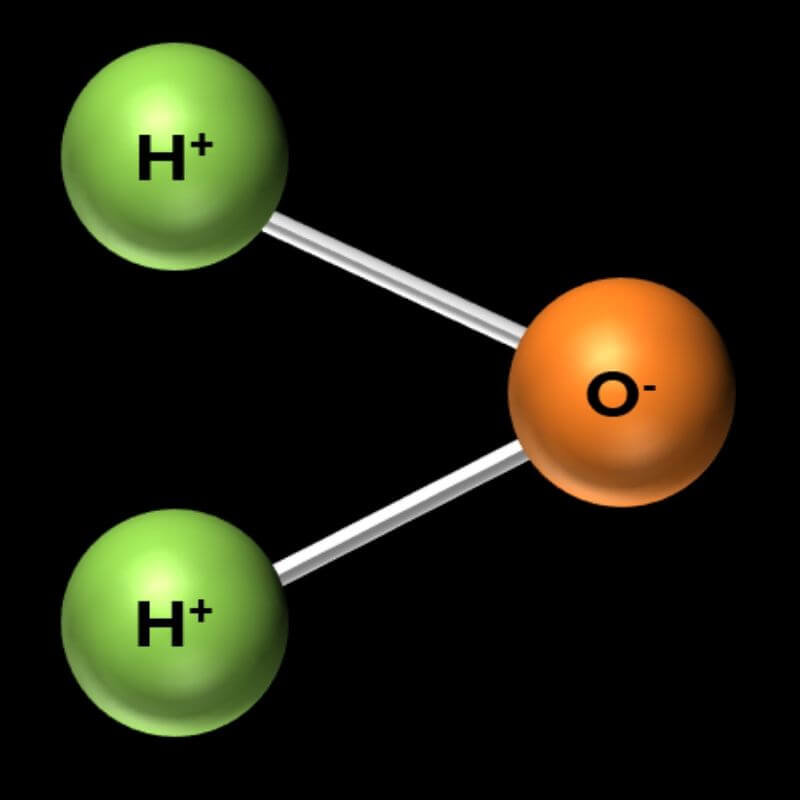
Water (H2O – Dihydrogen Monoxide):
- The most important molecule for life on Earth is H2O, or water. It is made of 2 hydrogen atoms and 1 oxygen atom.
- To make a water molecule, grab 2 marshmallows of one color and 1 of another. Then connect them with toothpicks, as shown in the picture. They should make a V shape.
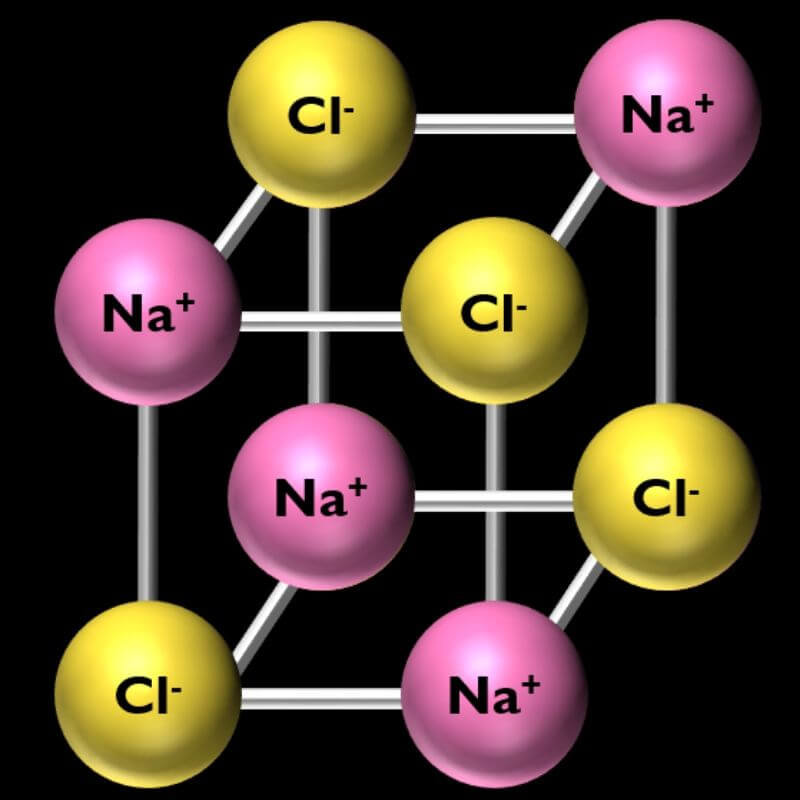
Salt (NaCl – Sodium Chloride):
- Salt molecules form cube-shaped crystals.
- To make a salt molecule, you will need 8 marshmallows total, 4 of one color, and 4 of another. Connect them together in a cube, as shown in the picture
Expand on this activity!
What other molecules can you make? Can you make methane? What about hydrogen peroxide? What’s the biggest molecule you can make? Check out MolView to see the digital models of all kinds of substances that you can base your marshmallow models off of!
Did you make your own marshmallow atomic models? We’d love to see how they turned out! Snap a photo of your models and submit it to our Science Showcase or tag Orlando Science Center and use #OSCatHome on social media! You might be featured on our channels.
The Science:
- Real molecules aren’t held together by toothpicks. Instead, the atoms are bound together by positive and negative charges.
- Water molecules are held together by covalent bonds, meaning they share negatively-charged particles called electrons.
- Salt is a different kind of molecule, one that is made of ions. This happens when an atom gains or loses an electron. Sodium (Na) loves to get rid of electrons, so it is usually positive. Chloride (Cl) loves to steal electrons, so it is usually negative.
- Molecules like this do not share electrons like water molecules do with covalent bonds. Instead, one atom gives an electron to the other, resulting in two charged atoms (ions). Just like with magnets, opposites attract. So, the positive sodium atoms and the negative chloride atoms will group together in the pattern that you’ve made. We call this an ionic bond.
OSC At Home Emails
Get a round up of our latest activities and ideas delivered straight to your inbox so you don't miss a thing!
Support OSC At Home
In these ever-changing times, it is our pleasure to adapt quality Orlando Science Center experiences to engage with everyone while they are safe at home. Please consider supporting our operating fund to ensure we can continue developing resources today and well into the future. Thank you for your generosity and support!

